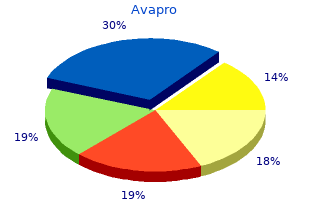
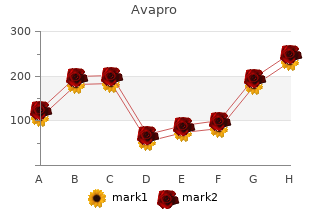
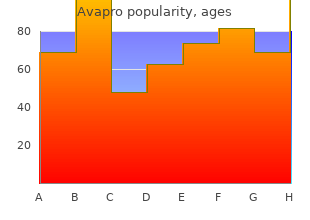
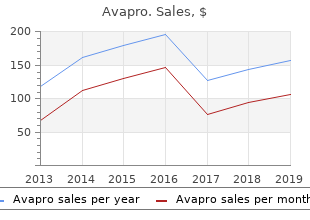
Avapro
"Generic avapro 300 mg, diabetic jelly beans".
By: M. Trompok, M.B. B.CH. B.A.O., Ph.D.
Co-Director, Rush Medical College
Also available in capsules that contain a viscous liquid ranging from 13 to diabetes okra order generic avapro online 150 mcg per capsule blood glucose monitor japan discount 150mg avapro. Capsules cannot be crushed diabetic compression socks buy avapro 150 mg line, suspended in water, or dissolved by placing in water before use. Stability tests demonstrated a 12% decline in levothyroxine concentration in the prepared suspension over 11 days. An oral liquid formulation of levothyroxine sodium 25 mcg/mL in 40% glycerol compounded from crushed tablets and distilled water with no preservatives added was stable for 8 days when stored in amber bottles at 4 degrees C. Title Levothyroxine Dose Initial oral dose: 10 to 14 mcg/kg/dose orally every 24 hours. Adverse Effects Prolonged over-treatment can produce premature craniosynostosis and acceleration of bone age. Monitoring After 2 weeks of treatment, serum levothyroxine (T4) concentration should be in the high normal range (10 to 16 mcg/dL) and should be maintained in this range for the first year of life. Special Considerations/Preparation 477 Micormedex NeoFax Essentials 2014 Oral suspension is not commercially available. Monitor patient closely when switching brand of drug due to some differences in bioavailability. To prepare a 15-mcg/mL levothyroxine oral suspension: Crush levothyroxine 100-mcg tablets in glycerol and add sterile water up to desired volume. Injectable form is available as lyophilized powder in vials containing 100 or 500 mcg. Product Information: levothyroxine sodium intravenous injection, levothyroxine sodium intravenous injection. Contraindications/Precautions Contraindicated in complete heart block and wide complex tachycardia attributable to accessory conduction pathways [3] [4]. Free drug fraction in both term and premature neonates is approximately twice that found in older children because of significantly reduced protein binding by I�1-acid glycoprotein. Later signs include seizures, loss of consciousness, respiratory depression, and apnea. Special Considerations/Preparation 480 Micormedex NeoFax Essentials 2014 Use only preservative-free lidocaine without epinephrine. Alteplase, aminophylline, amiodarone, ampicillin, caffeine citrate, calcium chloride, calcium gluconate, cefazolin, cefoxitin, chloramphenicol, cimetidine, dexamethasone, digoxin, dobutamine, dopamine, enalaprilat, erythromycin lactobionate, famotidine, fentanyl, flumazenil, furosemide, glycopyrrolate, heparin, hydrocortisone succinate, insulin, linezolid, methicillin, metoclopramide, micafungin, morphine, nafcillin, nicardipine, nitroglycerin, penicillin G, pentobarbital, potassium chloride, procainamide, ranitidine, sodium bicarbonate, and sodium nitroprusside. Title Lidocaine Antiarrhythmic Dose 481 Micormedex NeoFax Essentials 2014 Initial bolus dose: 0. Transformed in the liver to metabolites with antiarrhythmic activity; approximately 30% is excreted unchanged in neonates. Cardiac toxicity is associated with excessive doses and includes bradycardia, hypotension, heart block, and cardiovascular collapse. Caution: Preterm newborns and term newborns undergoing hypothermia treatment are at risk for drug accumulation due to slower drug clearance. Uses Treatment of severe recurrent or prolonged seizures that do not respond to first-line therapies. The clinically effective dose of 6 mg/kg/hr will lead to accumulation of both lidocaine and metabolites within several hours. Free drug fraction in both term and premature neonates is approximately twice that found in older children because of significantly reduced protein binding by alpha 1-acid glycoprotein. Measuring blood concentrations is not clinically useful except when accumulation is suspected. Pharmacology the mode of action for lidocaine as an anticonvulsant drug is unknown. Special Considerations/Preparation Use only preservative-free lidocaine without epinephrine. Alteplase, aminophylline, amiodarone, ampicillin, caffeine citrate, calcium chloride, calcium gluconate, cefazolin, cefoxitin, chloramphenicol, cimetidine, dexamethasone, digoxin, dobutamine, dopamine, enalaprilat, erythromycin lactobionate, famotidine, fentanyl, flumazenil, furosemide, glycopyrrolate, heparin, hydrocortisone succinate, insulin, linezolid, metoclopramide, micafungin, morphine, nafcillin, nicardipine, nitroglycerin, penicillin G, pentobarbital, potassium chloride, procainamide, ranitidine, sodium bicarbonate, and sodium nitroprusside. References Shany E, Benzaqen O, Watemberg N: Comparison of continuous drip of midazolam or lidocaine in the treatment of intractable neonatal seizures. Compatibilities updated 7/2009 Dose & Administration and References updated 1/2009 Added 3/2006 1.
Scientific Review Officer blood glucose of 102 discount 300mg avapro free shipping, Office of Contact Person: Lyle Furr what can you eat on a diabetes diet buy avapro with a visa, Scientific Contact Person: Jose F diabetic lifestyle buy avapro now. Drug Abuse Special Emphasis Panel, Loan (Catalogue of Federal Domestic Assistance Name of Committee: National Institute on Repayment 2016. Addiction Research Programs, National Translational Avant-Garde Award for Time: 9:00 a. Research ��Center of Excellence�� Grant National Institute on Drug Abuse; Name of Committee: National Institute on Program (P50). Core Center of Excellence Grant Program individuals associated with the grant applications, the disclosure of which Name of Committee: National Institute on (P30). Program Analyst, Office of Federal Advisory Coast Guard stakeholders and to facilitate the Committee Policy. The Sciences; Notice of Closed Meeting the availability, in the docket, of a working group held multiple meetings policy letter which provides voluntary and provided recommendations on Pursuant to section 10(d) of the guidance for the training of deck officers minimum standards of competence, sea Federal Advisory Committee Act, as on vessels operating in polar waters. The group also developed the meeting will be closed to the mariners working in this specialized recommendations on how existing public in accordance with the polar environment. The grant applications and International Code for Ships Operating adopted the working group�s the discussions could disclose in Polar Waters (Polar Code). The draft recommendations, which provided the confidential trade secrets or commercial policy letter and voluntary guidance basis of the U. In response guidelines for deck officers on vessels Biomedical Research and Research Training, to the challenges faced by these vessels operating in polar waters. Customs and Border from the entity that it is accredited or performing any duty or being assigned Protection, Department of Homeland approved by the U. Border Protection to conduct the familiar with those duties and specific test or gauger service requested. Inspectorate America Sediment in Crude Oils and Fuel Oils by the Ex Accreditation and Approval of Corporation is approved for the traction Method Inspectorate America Corporation, As following gauging procedures for 27�08 D86. Maritime Measurement etry Inspectorate America Corporation has 27�48 D4052 Standard Test Method for Density and Relative Den been approved to gauge petroleum and Inspectorate America Corporation is sity of Liquids by Digital certain petroleum products and accredited for the following laboratory Density Meter accredited to test petroleum and certain analysis procedures and methods for petroleum products for customs petroleum and certain petroleum purposes for the next three years as of products set forth by the U. D4928 Standard Test Method for Water in Crude Oils by Coulometric Karl Fischer Titration. D473 Standard Test Method for Sediment in Crude Oils and Fuel Oils by the Extraction Method. D445 Standard Test Method for Kinematic Viscosity of Transparent and Opaque Liquids. D4294 Standard Test Method for Sulfur in Petroleum and Petroleum Products by Energy-Dispersive X-ray Fluores cence Spectrometry. D4052 Standard Test Method for Density and Relative Density of Liquids by Digital Density Meter. D1796 Standard Test Method for Water and Sediment in Fuel Oils by the Centrifuge Method. Anyone wishing to employ this entity entity that it is accredited or approved inquiries regarding the specific test or to conduct laboratory analyses and by the U. Customs and Border gauger service this entity is accredited gauger services should request and Protection to conduct the specific test or or approved to perform may be directed receive written assurances from the gauger service requested. Calculations Approved Gauger and Accredited Laboratories Manager, Laboratories and 17. Standard Test Method for Water in Petroleum Products and Bitu minous Materials by Distillation. Standard Test Method for Sediment in Crude Oils and Fuel Oils by the Extraction Method. Standard Test Method for Sulfur in Petroleum and Petroleum Prod ucts by Energy-Dispersive X-ray Fluorescence Spectrometry. Standard Test Method for Density and Relative Density of Liquids by Digital Density Meter. Standard Test Method for Water and Sediment in Middle Distillate Fuels by Centrifuge. Standard Test Method for Water and Sediment in Fuel Oils by the Centrifuge Method. Standard Test Method for Kinematic Viscosity of Transparent and Opaque Liquids (and Calculation of Dynamic Viscosity).
These programs may be offered through a variety of sources diabetes symptoms back pain discount avapro line, such as comprehensive cancer centers diabetes type 2 nursing interventions cheap avapro 150 mg online, advocacy organizations diabetes symptoms 14 year old discount 150mg avapro free shipping, or community-based organizations (Tesauro et al. Policies may be implemented at the national, state, and community levels to create an environment supportive of advancing cancer survivorship in the realm of public health. An example of an existing policy that is relevant to cancer survivorship is the Cancer Survivors� Bill of Rights� (Spingarn, 1999). The Bill was written by a cancer survivor for cancer survivors and denotes the shift in a survivor�s role from passive patienthood to proactivity (Leigh & Stovall, 1998). This document serves as an example of how an advocacy organization can advance policy in the realm of cancer survivorship. Exploring ways that public health policy can be developed to address the needs of cancer survivors is an important next step in action planning. To ensure that cancer survivorship innovations reach the people who need them most, states, territories, and tribal organizations need to build and maintain appropriate infrastructure. Sufficient scientific and programmatic infrastructure will enable health agencies to build the necessary coalitions and partnerships to translate research into public health programs, practices, and services for cancer survivors. Patient navigation is a tool that can be used to ensure that survivors understand their care and their process of care and enhance the delivery of optimum care. In these programs, health professionals or highly trained patient liaison representatives coordinate health care 44 A National Action Plan for Cancer Survivorship: Advancing Public Health Strategies for patients and assist them in navigating the health care system. Develop and disseminate public education programs that empower survivors to make informed decisions. Within the realm of cancer survivorship, there is much to learn about the best practices of programs that address needs for people living with, through, and beyond cancer. Specific strategies to achieve the goal of identifying and disseminating best practices for cancer survivorship include the following: � Establish quantifiable criteria to determine which programs are among the best practices for addressing cancer survivor needs. States launched these Programs in collaboration with private and not-for-profit entities to assure appropriate expertise and to maximize the impact of limited resources on cancer control efforts. Public health agencies are using this support to establish broad-based cancer coalitions, assess the burden of cancer, determine priorities for cancer prevention and control, and develop and implement comprehensive plans. Through these and other activities, work is under way to identify those efforts that are grounded in sound scientific knowledge, or are �evidence based. Too often, rigorous evidence is lacking upon which to recommend strategies and interventions to address important goals and objectives. Most states have included issues related to cancer survivorship in their plans but have not necessarily included efforts that are evidence-based or that address needs for each stage of living with, through, and beyond cancer. There is a need to identify evidence-based initiatives that can be systematically incorporated into state cancer control efforts. The following strategies provide specific guidance to meet this need: � Identify key leaders and experts in cancer survivorship in every state (especially survivors) to create a network of individuals to ensure that survivorship issues are being addressed through each cancer plan. These guidelines summarize the collective research on outcomes pertaining to one disease. Historically, cancer was a disease that people often did not survive (see Section I. Health care focused on making the patient comfortable during the last stages of cancer progression; few treatment options were available. Now, many more treatment options are available, and people survive with cancer for many years. The medical model tends to focus more on cancer survivors during their �acute� stages of cancer and less on the �extended� and �permanent� stages (Mullan, 1985) and not on post-treatment or long-term issues. Policies need to effectively address cancer survivorship for all those living with, through, and beyond cancer. Strategies for effecting this change include the following: � Develop and disseminate public education materials to educate policy makers, health professionals, and survivors on the stages of cancer survivorship. A great deal is unknown about cancer survivorship, particularly in terms of the long-term effects of cancer diagnosis and treatment. For that reason, much work needs to be done to create comprehensive databases to collect information on survivors and conduct research on issues related to survivorship. Develop, test, maintain, and promote patient navigation or case management programs that facilitate optimum care. Implement evidence-based cancer plans that include all stages of cancer survivorship. Promote policy changes that support addressing cancer as a long-term, chronic disease.
In the reactive near-field region diabetic polyneuropathy cheap generic avapro canada, there is no simple relationship between the E and H fields (the impedance (E/H) will differ from 377 ohms) managing diabetes during holidays buy discount avapro 300mg online. The linear decrease in field strength with distance and the decrease in power density with distance squared that is characteristic of the far field do not apply in the near field region diabetic diet eating out order 300mg avapro free shipping. For example, placing a probe near a source or standing near a source while carrying out measurements may change the characteristics of the fields considerably. These factors include: a) Variation of probe impedance with proximity to nearby reflective surfaces; b) Capacitive coupling between the probe and the field source; and c) Non-uniform illumination of the sensing elements that make up the probe. Maintaining adequate separation distance between the probe elements and the field source can eliminate the influence of each of these factors, which otherwise could result in erroneous field strength measurements. Accordingly, measurements should be made at a distance no closer than three probe-diameters between the center of the probe and any object, or 20 cm�whichever is greater. For practical measures of compliance with the standard, the average of a series of ten field strength measure ments performed in a vertical line with uniform spacing starting from about 20 cm above ground level up to a height of 2 m is deemed sufficient (spatial averaging). However, the measurement spacing should be held constant so as to obtain a true spatial average. However, non compliance with the spatial average or spatial peak values may be overturned by demonstrat ing compliance with the basic restrictions using other analytical methods. These are typically much more complex to obtain: so it is usually easier to first test for compliance by measurement. A maximum of five pulses with pulse durations less than 100 ms is permitted during any period equal to the averaging time. If there are more than five pulses during the averaging time, or if the pulse duration is more than 100 ms, normal averaging time calculations apply. But, 2 2 2 Stdt = (1 200 000 W/m)(10 ms) = 12 000 J/m >7200 J/m Although the exposure meets the peak electric-field strength criterion it does not meet the energy density cri terion and, therefore, does not meet the peak power limitations for the controlled environment. In each example, the pulse width was significantly longer than the time between each complete oscillation of the microwave frequency. In this section, an example of how to assess compliance when the pulse is a non-sinusoidal waveform is provided. The phase duration tp (defined as the time between zero crossings of a waveform having zero mean) of this pulse is 0. Then test for compliance with either the peak field restriction or the Fourier component restriction. The external B and E field strengths are limited by the pulse rise time (since real pulses are never square) expressed as the time rate of change of the B or E field (dB/dt or dE/dt). In this example, the fundamental frequency component is 10 kHz, the 3rd harmonic is 30 kHz, the 5th is 50 kHz, the 7th is 70 kHz, the 9th is 90 kHz, the 11th is 110 kHz, and the 13th is 130 kHz. In this controlled environment application, the squares of the peak values observed (81 V/m and 0. The values shown in columns 2, 3, and 6 in the table below represent the measured frequency and the electric and magnetic field strengths as averaged over an area equivalent to the vertical cross section of an adult. The far field of a simple antenna is generally defined as starting at a distance of five wavelengths from the antenna or, in the case of an antenna with a parabolic reflector, at a distance of ten times the diameter of the reflector. For an antenna with multiple elements, the radiation pattern of the antenna can be considered to be fully formed at a distance of ten times the maximum element spacing. Accurate depiction of exposure to determine compliance with exposure standards therefore requires the measurement of both field components. Absent a focusing effect (which might be produced by a pair of orthogonally-related conduct ing surfaces), the total absorbed energy is no greater than would be experienced in the absence of the reflecting object. In addition, locations where the distribution of electromagnetic energy exhibits a complex pattern, compliance with the pertinent standard for maximum permitted exposure may be better determined by measuring the induced current in the subject than by measuring field strength. This condition may occur particularly in locations where multiple emitters, utilizing a variety of frequencies and at different locations, are producing the total exposure environment. Intuitively, one may conclude that, at some level of electric field strength, induced currents in the human body cannot exceed the standard, thus making unnecessary current measurements to show compliance with the standard. Results of those calculations have been included in the standard as percent of maximum permitted electric field strength versus frequency.
Discount avapro 150mg. Red Light Therapy for Diabetes and Insulin Resistance.

