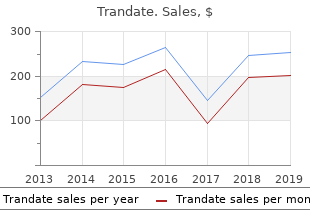
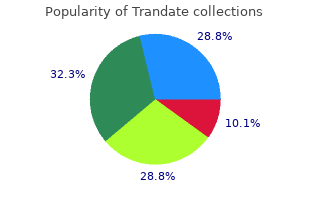
Trandate
"Buy discount trandate 100mg, pulse pressure and map".
By: E. Pakwan, M.B. B.CH. B.A.O., M.B.B.Ch., Ph.D.
Vice Chair, Marian University College of Osteopathic Medicine
References of the identified articles were searched for voltage-gated potassium channels blood pressure 3060 order genuine trandate. Dev Med Child Neurol 2015;57: romyotonia: autoimmune pathogenesis and response to heart attack 26 effective 100 mg trandate immune 431�440 blood pressure ranges american heart association order 100mg trandate otc. Effect of rituximab in patients with leucine-rich, glioma Neurology 2008;70:1883�1890. Muscle Nerve in patients with neurologic symptoms due to antibodies to 1994;17:S221. J Neurol myotonia and limbic encephalitis sera target mature Shaker 1999;246:623�625. Children present with asymotomatic liver deposits of copper, teenagers with liver disease, and adults with neurological symptoms. History of behavioral disturbances is present in half of patients with neurological disease. Zinc acetate is nontoxic and stimulates metallothioneine which reduces dietary and enterohepatic absorption of copper. It is the therapy of choice for asymptomatic patients or patients with hepatitis or cirrhosis, but without evidence of hepatic decompensation or neurologic/psychiatric symptoms. Decreased serum copper may decrease hemolysis, prevent pro gression of renal failure, and provide clinical stabilization. Plasma/albumin combination is also possible as use of albumin alone will worsen coagulopathy. Motobayashi M, Fukuyama T, Nakayama Y, Sano K, Noda S, Hidaka Y, Amano Y, Ikeda S, Koike K, Inaba Y. Nagata Y, Uto H, Hasuike S, Ido A, Hayashi K, Eto T, Hamakawa for additional cases and trials. Narumi S, Umehara M, Toyoki Y, Ishido K, Kudo D, Kimura exchange effective in prevention of hepatic transplantation in N, Kobayashi T, Sugai M, Hakamada K. J Pediatr Gastro for Wilson�s disease in pediatric patients: decision making and enterol Nutr 2011;52:778�780. Wilson�s disease and other plantation in Wilson�s disease based on the outcomes of patients neurological copper disorders. Anaesth Inten plasmapheresis as a bridge to liver transplantation in fulminant sive Care 2013;41:811�812. Interessenskonflikt Alle beteiligten AutorInnen erklaren, dass keine Interessenskonflikte im Sinne der Uniform Requirements of Manuscripts Statement of Medical Journal Editors ( Fruhzeitige Informationen fur gesundheitspolitische Informationen uber Entscheidungstrager daruber, welche Interventionen (Impfungen und *Status von F&E Medikamente) derzeit in klinischen Studien erprobt werden und *evidenzbasierter b. Monitoring dieser in den nachsten Monaten, um gegebenenfalls einen Einkauf evidenzbasierten Einkauf zu unterstutzen. In einem zweiten Schritt wird eine selektive Recherche mittels Monitoring Handsuche in Studienregistern nach Informationen uber klinische Studien am Menschen und den Stand der Forschung durchgefuhrt. Diese Informationen bilden die Grundlage fur sogenannte "Vignetten" (Kurzbeschreibungen) fur jene Produkte, die sich bereits in einem "fortgeschrittenen" Stadium befinden. Anschlieend werden die Produkte hinsichtlich des Status der klinischen Studien bis zur Zulassung beobachtet und schlielich auf Nutzen und Schaden bewertet. Alle Arbeitsschritte werden in enger internationaler (europaischer) internationale/ Zusammenarbeit durchgefuhrt. Zusammenarbeit Zusatzlich werden Informationen uber offentliche Mittel fur die Entwicklung zusatzlich: offentliche von Medikamenten und Impfstoffen gesammelt. Finanzierung von F&E Folglich wird der vorliegende Bericht mehreren Versionen verfugbar sein: V1: Bestandsaufnahme � Version 1 (V1, April 2020): Bestandsaufnahme + Vignetten fur die bis + Vignetten dato fortgeschrittensten Produkte V2: monatliches � Version 2+ (V2, V3): monatliches Monitoring und Update Monitoring 8 Table 0-1. Beschreibung in Form "fortgeschrittenen" von �Vignetten") ausgewahlt: Stadien � Produkte, die sich bereits in einem �fortgeschrittenen" Stadium haufig genannte befinden, potentielle Kandidaten � Produkte, die am haufigsten in klinischen Fachzeitschriften als potenzielle Kandidaten diskutiert werden. Bei einem 19 Groteil der Wirkstoffe, die derzeit getestet werden, handelt es sich um �Repurposing�, also Medikamente, die bereits fur andere Indikationen Repurposing (vorrangig andere virale Infektionen) zugelassen sind.
Treatmentof ointmentform) th e patientwith anterioruveitismay require consultationwith orreferral � M edrysone 1% blood pressure empty chart buy cheap trandate 100mg. B asisfor T reatment A llcycloplegicagentsare ch olinergicantagonistswh ich work by blockingneurotransmissionatth e receptorsite ofth e irissph incterand Th e generalgoalsforth erapy inanterioruveitisare: ciliary muscle blood pressure chart metric purchase trandate 100mg. Th e treatmentofanterioruveitisisnonspecific blood pressure medication zanidip purchase 100 mg trandate with mastercard,usually involvingtopical th erapy with corticosteroidsand cycloplegics. Th e role ofcorticosteroidsintreatinganterioruveitisisto decrease Ph enyleph rine,2. B ecause ph enyleph rine h as membranes,inh ibitingth e release oflysozyme by granulocytes,and neith eracycloplegicnoranti-inflammatory effectand may cause a 26 release ofpigmentcellsinto th e anteriorch amber,itisgenerally not suppressingth e circulationoflymph ocytes. O ralSteroidsandN onsteroidalAnti-InflammatoryDrugs � R eferral: Th e optometristsh ould considerreferringth e patientto th e appropriate ph ysicianforsystemicworkupwh enth ere is O ralprednisone may be utilized inrecalcitrantcasesofanterioruveitisin recurrence orbilateralinvolvement(Table 2). O th erTh erapies W h entopicalth erapy fails,perioculardepotinjectionsare useful. M ild M oderate Severe Immunosuppressive th erapy representsa"lastresort"inth e treatmentof anterioruveitis. Patientsrequiringsuch th erapiesmay require M ildto moderatesymptoms M oderateto severesymptoms M oderateto severesymptoms consultationwith anoph th almologist. R ecommendedTh erapeuticR egimen* Superficialcircumcornealflush Deepcircumcornealflush Deepcircumcornealflush Th e initialtreatmentofanterioruveitismay include th e use of: N o K Ps ScatteredK Ps DenseK Ps Traceto 1+ cellsandflare 1-3+ cellsandflare 3-4+ cellsandflare � C ycloplegicagents: C yclopentolate,1%,may be used th ree times M iotic,sluggish pupil Sluggish orfixedpupil perday (t. TheCareProcess25 26AnteriorU veitis Th e initialfollowupofanterioruveitispatientssh ould be sch eduled T able5 between1-7 days,dependingonseverity. R eferto primary care ph ysicianforsystemicevaluation(wh en Ifatfollowup,th e anteriorch amberreactionisimproving,th e clinician indicated) may continue orreduce medications,dependingonth e severity ofth e initialreaction. Duringtapering,th e cliniciansh ould observe th e patientforsignsofincreased inflammation. C ompliance with th e th erapeuticregimenand keepingall * A daptedfrom C ataniaL J. N orwalk,C T: followupappointmentsare essentialto ach ieve th e th erapeuticgoals. TheCareProcess27 Th e optometristsh ould advise th e patientofth e potentialside effectsof long-term corticosteroid use. Itisimportantth atth isadvice be well-documented inth e medicalrecord,and th e patientsh ould be reminded periodically th rough outth e course oftreatment. Prognosis M ostcasesofanterioruveitisrespond favorably to early diagnosisand treatment. Th erefore,both th e clinicianand patientmustbe alert forsignsofrecurrence and reinstitute th erapy promptly. C linicalfeatures Th e optometristplaysanimportantrole inth e ongoingcare ofth e patient ofacute anterioruveitis. U veitis: many diseases,one cases,regularoptometricexaminationsare essentialto preservingeye diagnosis. O ph th almologicalscreeningin juvenile arth ritis:sh ould th e frequency ofscreeningbe based on th e risk ofdevelopingch roniciridocyclitis Efficacy and safety of rimexolone 1% oph th almicsuspensionvs1% prednisolone acetate 17. Signsand symptomsof ch ildh ood iridocyclitiswith nonsteroidalanti-inflammatory drugs. W B C W h ite blood cells Appendix39 40AnteriorU veitis M ydriasis Dilationofth e pupil. O ph th almoscopy Examinationofth e interiorofth e eye,usingan illuminationsystem involvingth e ligh tsource,lenses,and aprism or V augh anD,A sbury T,R iordon-EvaP. Posterior subcapsular cataractL ossoftransparency ofth e eye orits capsule involvingth e cortexatth e posteriorofth e pole ofth e lens. Synech ia A nadh esionofth e iristo th e cornea(periph eralanterior synech ia)orlens(posteriorsynech ia). V isualacuity Th e clearnessofvisionth atdependsuponth e sh arpness offocusofth e retinalimage and th e integrity ofth e retinaand visual path way.
Buy trandate 100 mg low cost. L-Arginine For High Blood Pressure - http://www.detorex.com.
Persistent pruritic papules and plaques with scale and linear pigmentation (Correct) B heart attack quizzes 100mg trandate for sale. Evanescent non-pruritic non-scaly salmon-colored morbilliform eruption (Correct) C arteria epigastrica cranialis superficialis commissura labiorum dorsalis order 100 mg trandate overnight delivery. Intermittent and recurrent urticarial eruption with non-pruritic erythematous macules or slightly elevated plaques (Correct) D blood pressure xanax discount trandate 100mg on-line. All of the above (Correct) Discussion Adult-onset Still�s disease is a rare systemic inflammatory disorder of unknown etiology that was first described by Eric Bywaters in 1971 in a report detailing 14 adult patients who presented with arthritis and systemic features similar to those seen in systemic juvenile-onset rheumatoid arthritis (systemic juvenile idiopathic arthritis Still�s disease). There are no pathognomonic signs and symptoms of adult-onset Still�s disease and the clinical course is variable, often resulting in delay in diagnosis and treatment. Although several diagnostic criteria have been proposed, the most commonly used and best validated is the Yamaguchi classification which requires five or more criteria, including two or more major criteria and exclusion of infections, malignancies, and other rheumatic diseases. Although the non-scaly non-pruritic intermittent salmon-colored maculopapular rash is typically the initial cutaneous manifestation seen, there are a variety of other skin eruptions that can initially present or subsequently develop in adult-onset Still�s disease, as described above in question 2. Cutaneous eruptions have shown to change with ongoing disease with transition, most commonly, from the classical morbilliform salmon colored rash to persistent hyperpigmented plaques with a rippled or linear appearance. Histological features: 256 � Classical rash= subtle perivascular and interstitial inflammation in the superficial dermis consisting of lymphocytes and scattered neutrophils. Dermal changes include a superficial perivascular infiltrate of lymphocytes and neutrophils and an increase in interstitial dermal mucin. Yamaguchi M, Ohta A, Tsunematsu T, Kasukawa R, Mizushima Y, Kashiwagi H, Kashiwazaki S, Tanimoto K, Matsumoto Y, Ota T. Question the best diagnosis is: A) Deep penetrating nevus (Incorrect) Deep penetrating nevi have a sharply demarcated, circumscribed, wedge-shaped architecture with a limited junctional component and epithelioid dermal melanocytes with abundant eosinophilic or amphophilic cytoplasm arranged in a plexiform pattern as loose nests and vertically oriented fascicles with discohesion at the periphery and base. The melanocytes extend down along adnexal structures into the deep dermis and subcutis and do not show obvious maturation. Perineural extension and involvement of the arrector pili muscles are frequently seen. B) Melanoma (Incorrect) Features frequently seen in melanoma are listed in question 2 and include epithelioid cells with striking pleomorphism and large and/or multiple nucleoli, infiltrative borders, frequent associated inflammation, abundant (>3 /mm2) and/or atypical mitoses, necrosis, lymphovascular invasion, and often the presence of a junctional component. C) Cellular neurothekeoma (Incorrect) Cellular neurothekeoma has an ill-defined plexiform or multi-lobular architecture and is composed of fascicles, nests, and whorls of epithelioid or spindled cells with ample eosinophilic cytoplasm and monomorphous nuclei. D) Cellular blue nevus (Correct) Cellular blue nevus has a well circumscribed, nodular to dumb-bell shaped architecture with a biphasic pattern consisting of classic or common blue nevus areas and distinct hypercellular areas, particularly in the deeper portions of the lesion. The hypercellular areas form nests, nodules, fascicles, and alveolar patterns and are composed of spindled cells with monomorphous nuclei, even chromatin, inconspicuous nucleoli, and moderate amphophilic to lightly pigmented cytoplasm. Degenerative changes, including hemorrhage, cystic or myxoid areas, fibrosis, and stromal hyalinization are often present. E) Angiosarcoma (Incorrect) Angiosarcomas are composed of irregular anastomosing blood vessels that dissect collagen bundles throughout the dermis. The neoplastic endothelial cells can be epithelioid and have multi-layering, nuclear pleomorphism, and mitoses. An associated inflammatory infiltrate, extravasated red blood cells, and hemosiderin are usually present. In contrast to the features listed below, histopathological changes that support cellular blue nevus over melanoma include absent junctional activity, pushing well circumscribed borders with a nodular or dumb-bell shape architecture, absence of associated inflammation, biphasic pattern with areas of common blue nevus associated with areas of hypercellularity, fasciculation, spindled rather than epithelioid cytology, lack of significant cellular pleomorphism, rare and typical mitoses (1 /mm2), single and small nucleoli, absence of necrosis, and infrequent ulceration. All of the above (Correct) Discussion Cellular blue nevus was originally thought to be a variant of melanoma (so-called �melanosarcoma�), but Allen, in 1949, was the first to recognize it as a benign cellular variant of blue nevus. Cellular blue nevi predominately occur in Caucasian females between the ages of 10-40 years old, and are most commonly found on the buttock or sacrococcygeal region, scalp or face, proximal extremities, and trunk. Clinically, cellular blue nevi are blue, blue-black, or black firm to rubbery dome-shaped solitary nodules with smooth borders. A reorientation on the histogenesis and clinical significance of cutaneous nevi and melanomas. Fluorescence in situ hybridization for distinguishing cellular blue nevi from blue nevus-like melanoma. The patient does not have a known history of pancreatitis or other pancreatic disorder. During his hospitalization, multiple cultures were taken from the subcutaneous lesions which were negative.
Which of the following types of units do you customarily use to blood pressure yoga ramdev order cheap trandate describe marketed drug use Although your answer may depend on the particular source used for denominator data blood pressure medication diabetes generic trandate 100 mg visa, indicate the best match (narrowest gap) blood pressure medication pregnancy order 100 mg trandate visa. Who in your organization actually determines/derives the estimated ��denominator�� for marketed drugs for use in clinical safety applications Company Regulator 15 4 Safety/epidemiology staff 7 0 Marketing research staff 1 0 Clinical department 0 0 Biometrics department � � Other (specify)* * Company comments on other. When needed, are you generally able to obtain dissection of exposure data broken down by: Company Regulator Breakdown Category 15 4 Location of use. Company Regulator 5 2 Same as for prescription products 0 0 Special techniques � � Explain* * Company comment. This Question Applies Only To Manufacturers In most companies, the marketing research departments compile data from sales statistics and other sources. How would you describe the relationship between the safety/epidemiology and marketing research opera tions with regard to provision of needed exposure data Circumstances Surrounding the Determination of Exposure Data Depending on circumstances and your practice, for the submission of a periodic safety report on a marketed drug involving no known major safety (or efficacy) issues, it may be sufficient to provide a gross, overall estimate of the exposed population. However, in the event of a safety problem, for example, it may be necessary or useful to attempt a finer dissection of the data (see items in question B. Do you draw such a distinction between these two circumstances or do you attempt to obtain detailed breakdowns of exposure data routinely As part of ongoing safety monitoring and assessment, it might be possible to use different levels of exposure data to evaluate various 343 aspects of a product�s safety profile. For each of the following sample ��scenarios,�� indicate whether you routinely perform the indicated assessments, on either a periodic (interval) or cumulative data basis Evaluate patient exposure by age and/or gender to determine whether the benefit-risk relationship is different (or has changed) for special populations. Comparison of use of a drug alone to the drug in combination with specific concomitant therapy in order to evaluate the possibility of drug interactions. The purpose of this question is to determine the nature of ��denomi nator�� data you receive or obtain yourself and your perspective on the adequacy of those data for pharmacovigilance purposes. It is recognized that the quantity and quality of such data may vary greatly from circumstance to circumstance, even company to company. In general, how would you describe the type and amount of exposure data you receive in manufacturers� periodic safety reports: Excellent Good Fair Poor No Response Regulator 2. In general, how would you describe the manufacturers� use and interpretation of the exposure data they provide. Briefly describe the principle improvements you would like to see in the types, amounts, and uses of exposure data submitted by manufacturers. Also, consideration of how a product is used and in what populations, when assessing safety profiles of a drug. If you make your own estimations of exposure data, independent of manufacturers, please describe what sources and methods you use for (a) routine, general purposes and (b) for special safety problem situations. Comments Please provide whatever questions, suggestions and concerns you believe are important in addressing the issue of denominator determination and application. Any special situations for which you have experience would be particularly helpful. For example: exposure data in developing countries; data on drugs that are used in two or more distinct indications or populations (pediatrics vs adults, different dosage forms, etc. Company comments: Additional estimates of exposure should be provided for o Known risk populations o Children o Off-label use Estimation of denominators remains a challenging area. In general, it would seem that only Pharmacovigilance is interested in patient denominator data. Other departments request this information from us (although we don�t have access to the raw data) to put their data into context. Also the data obtained this way are at significant variance with our estimates using kg sold or unit doses sold. For spontaneous report data the numerator is terrible, but the denominator is clearly worse. In collaboration with sales and network staff, developed a program that extracts units sold on a monthly basis by code which combines all 347 formulations of a product and factors in a ��constant�� to correct for how the raw data are input into a master table.

